Local TH action in acute and chronic ischaemic heart disease
Prof. Dr. med. PhD Dagmar Führer, Prof. Dr. med. Lars C. Möller & Prof. Dr. Kristina Lorenz
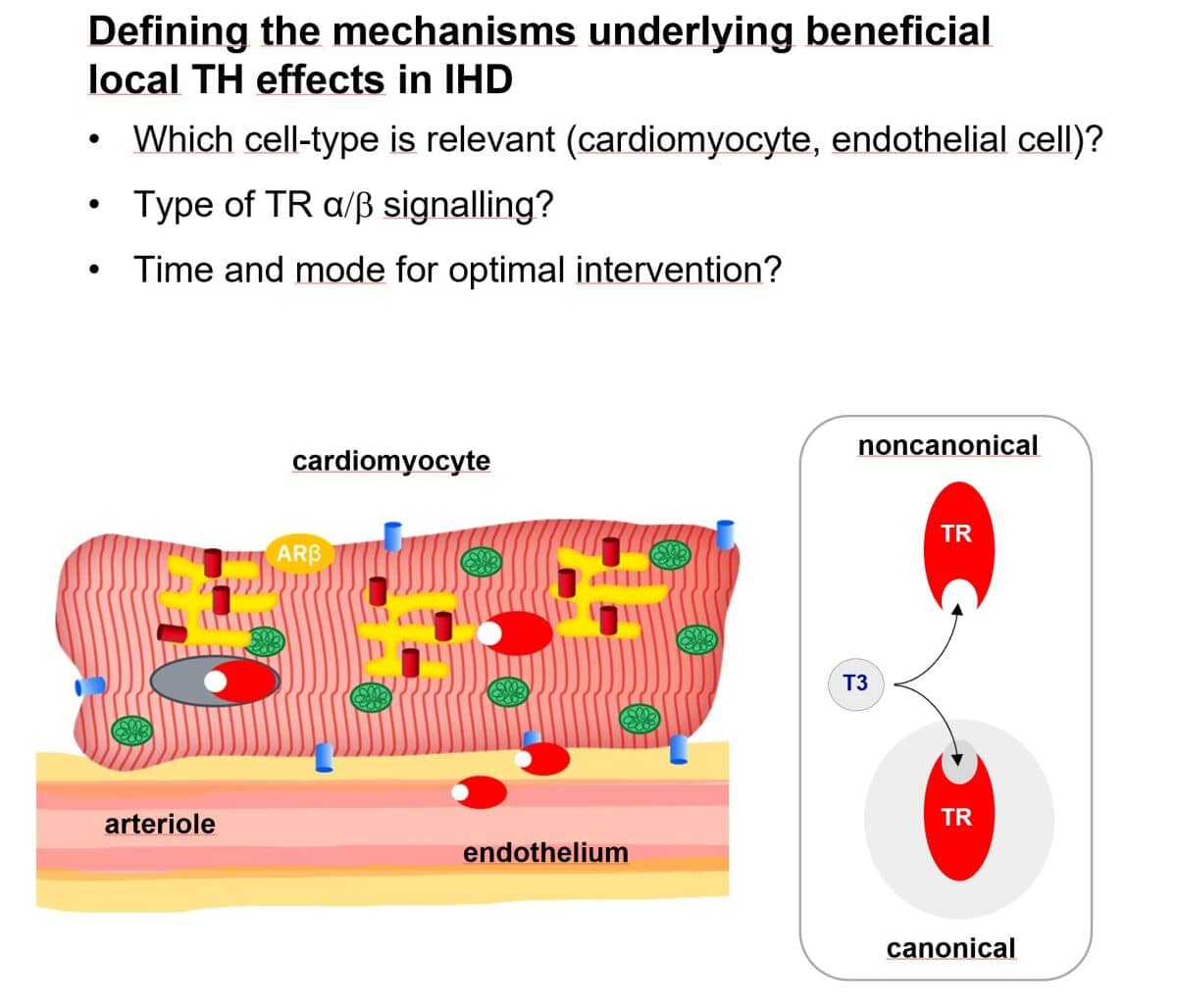
This project pursues the hypothesis that targeted TH action can be applied to reduce injury in acute myocardial infarction (MI) and to ameliorate post-MI remodelling. Using cell-specific mouse models, we will comprehensively characterise local control of TH action in acute and chronic ischaemic heart disease (IHD) and in these conditions will dissect the role of TR isoform, and the mode of TR action in endothelial cells and cardiomyocytes. Furthermore, we will define the optimal timing for TH intervention and will explore selective targeting. Our long-term goal is to exploit local TH action for the benefit of patients with IHD.
Further Information
Selected Publications
Noncanonical Action of Thyroid Hormone Receptors α and β.
Hönes GS, Geist D, Moeller LC.
2020. Exp Clin Endocrinol Diabetes 128(6-07):383-387. doi: 10.1055/a-1088-1187
Increased Anaplastic Lymphoma Kinase Activity Induces a Poorly Differentiated Thyroid Carcinoma in Mice.
Kohler H, Latteyer S, Hönes GS, Theurer S, Liao XH, Christoph S, Zwanziger D, Schulte JH, Kero J, Undeutsch H, Refetoff S, Schmid KW, Führer D, Moeller LC.
2019. Thyroid 29(10):1438-1446. doi: 10.1089/thy.2018.0526
Thyroxine promotes lung cancer growth in an orthotopic mouse model.
Latteyer S, Christoph S, Theurer S, Hönes GS, Schmid KW, Führer D, Moeller LC.
2019. Endocr Relat Cancer 26(6):565-574. doi: 10.1530/ERC-18-0353
Noncanonical thyroid hormone signaling mediates cardiometabolic effects in vivo.
Hönes GS, Rakov H, Logan J, Liao XH, Werbenko E, Pollard AS, Præstholm SM, Siersbæk MS, Rijntjes E, Gassen J, Latteyer S, Engels K, Strucksberg KH, Kleinbongard P, Zwanziger D, Rozman J, Gailus-Durner V, Fuchs H, Hrabě de Angelis M, Klein-Hitpass L, Köhrle J, Armstrong DL, Grøntved L, Bassett JHD, Williams GR, Refetoff S, Führer D, Moeller LC.
2017. Proc Natl Acad Sci U S A 26;114(52):E11323-E11332. doi: 10.1073/pnas.1706801115
A 6-Base Pair in Frame Germline Deletion in Exon 7 Of RET Leads to Increased RET Phosphorylation, ERK Activation, and MEN2A.
Latteyer S, Klein-Hitpass L, Khandanpour C, Zwanziger D, Poeppel TD, Schmid KW, Führer D, Moeller LC.
2016. J Clin Endocrinol Metab 101(3):1016-22. doi: 10.1210/jc.2015-2948
A new type of ERK1/2 autophosphorylation causes cardiac hypertrophy.
Lorenz K, Schmitt JP, Schmitteckert EM, Lohse MJ.
2009. Nat Med 15(1):75-83. doi: 10.1038/nm.1893
Interference with ERK-dimerization at the nucleocytosolic interface targets pathological ERK1/2 signaling without cardiotoxic side-effects.
Tomasovic T, Brand T, Schanbacher C, Kramer S, Hümmert MW, Godoy P, Schmidt-Heck W, Nordbeck P, Ludwig J, Homann S, Wiegering A, Shaykhutdinov T, Kratz C, Knüchel R, Müller-Hermelink HK, Rosenwald A, Frey N, Eichler J, Dobrev D, El-Armouche A, Hengstler JG, Müller OJ, Hinrichs K, Cuello F, Zernecke A, Lorenz K.
2020. Nat Commun 11, 1733. doi: 10.1038/s41467-020-15505-4
Phosphodiesterase 2 Protects Against Catecholamine-Induced Arrhythmia and Preserves Contractile Function After Myocardial Infarction.
Vettel C, Lindner M, Dewenter M, Lorenz K, Schanbacher C, Riedel M, Lämmle S, Meinecke S, Mason FE, Sossalla S, Geerts A, Hoffmann M, Wunder F, Brunner FJ, Wieland T, Mehel H, Karam S, Lechêne P, Leroy J, Vandecasteele G, Wagner M, Fischmeister R, El-Armouche A.
2017. Circ Res 120(1):120-132. doi: 10.1161/CIRCRESAHA.116.310069
Interference with ERKThr188-phosphorylation impairs pathological but not physiological cardiac hypertrophy.
Ruppert C, Deiss K, Herrmann S, Vidal M, Oezkur M, Gorski A, Weidemann F, Lohse MJ, Lorenz K.
2013. Proc Natl Acad Sci U S A 110(18):7440-5. doi: 10.1073/pnas.1221999110
Cardiac RKIP causes a beneficial beta-adrenoceptor-dependent positive inotropy.
Schmid E, Neef S, Berlin C, Tomasovic A, Kahlert K, Nordbeck P, Deiss K, Denzinger S, Herrmann S, Wettwer E, Weidendorfer M, Becker D, Schäfer F, Wagner N, Ergün S, Schmitt JP, Katus HA, Weidemann F, Ravens U, Maack C, Hein L, Ertl G, Müller OJ, Maier LS, Lohse MJ, Lorenz K.
2015. Nat Med 21(11):1298-306. doi: 10.1038/nm.3972







